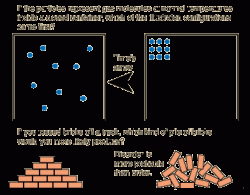
If you want an intuitive way of thinking about what the Boltzmann entropy $S = k \log \Omega$ tells us, instead of disorder, I would say that entropy is a measure of ignorance. So unless you have some idea already in mind of what you mean by order, I would avoid thinking about entropy in those terms. You could just declare that disorder is quantified by entropy, but I find that our intuitive notion of disorder doesn't always agree with what we get when we calculate the entropy. In general, "order" and "disorder" are not really well-defined or quantifiable. So, if you want to retain the use of the words order and disorder, you should assume that order refers to conditions in which a relatively large amount of energy is possessed by a relatively small number of particles within a system, while disorder refers to conditions in which the energy is more randomly dispersed among the population of particles. In principle it is possible for the particles to interact with each other so that once again the energy becomes more concentrated among a tiny fraction of them, but the odds of that happening are vanishingly small. With increasing time a sort of equilibrium's reached in an isolated system, at which the energy is randomly distributed among the particles. The effect is that the high level of energy initially focused in a single particle is gradually diffused among all the particles. At first, it has a lot of energy to pass-on to any particle with which it interacts, so it spawns, as it were, other particles with more energy than the rest, and they in turn interact with other particles and so on in a cascade. If you have one very energetic particle which subsequently interacts with many others with less energy, it tends to lose more and more of its energy with each interaction. Whenever particles interact, energy passes between them. It is better to think of increasing entropy as meaning the averaging out of energy among multiple particles. The use of the terms order and disorder in descriptions of entropy can be the source of confusion, and is unnecessary. The more disordered a state is, the more ways that state can be achieved, and the more probable it is that it will occur. But there's a million ways for the milk to become dispersed into the coffee and we have no idea which one is actually going to happen, so it is the more disordered and far more likely state. If you pour milk into coffee, the system would be well ordered if the milk and coffee didn't mix: there's only one way for every atom to be exactly where it was put. On the macroscopic scale, disorder tends to be related to homogeneity. If the sum was something like 21, this would be very disordered (135246 or 635241 or 333444 or 526152 or.). So you know most of the dice are 6, but each one has a 1/6 chance of actually being a 5. 
If you have a total of 35, this is slightly less ordered: one of the dice is a 5, and it could be any of them. This is a well ordered state, you know exactly what's going on with every die. If you roll 6 dice, there's only one way you can get a total of 36: every die is a 6. If entropy is a measure of disorder, then 'order' is also related to the number of microstates-the number of different ways a system can be in a given state. So we see china plates shatter into lots of pieces when dropped on a concrete floor, but we never see those pieces spontaneously jump into the air and re-form themselves into an unbroken plate.Įntropy is dependent on the number of microstates. 
In our universe, systems always tend naturally towards states of greater entropy as time goes by. The random deck of cards has high entropy, which becomes low entropy when you sort them out again.īut that entropy reduction required the expenditure of work, which produced an increase of entropy somewhere else in the process of furnishing that work. The sorted deck has low entropy, which becomes high entropy when you shuffle them randomly. If you dropped a whole randomly-shuffled deck of cards onto the floor and then scooped them all up, it is extremely unlikely that they would assemble themselves into that sorted order.įurthermore if you dropped the sorted deck onto the floor, it is extremely likely that after you scooped up all the cards you would find them all scrambled up, out of order. This deck has a lot of order built into it. Imagine a deck of playing cards in which each suit is sorted out and all the cards in each suit arranged in descending order. Here is a slightly different take on this.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
